Caught In A Big Lie
A Meditation on How South Africa's Finest Bankers Handle Arithmetic and Truth
In the Shadows of COVID-19
Investigative journalists are, by nature, incurable dot-connectors. Call it apophenia if you like; just don’t mistake it for conspiracy. At uSpiked, we plead guilty to pattern-spotting, not paranoia. We follow the data, and the…
Social Image Laundering
DON’T LOOK THERE! Just look at what we have here! Our solidarity is with our slayed colleagues in Gaza
Trade Wars
Elon Musk: As strange as it may seem, would anyone have predicted that it was only to take a Pretoria Boys’ Alumni to bring the mighty United States of America to its knees while wreaking havoc globally?
What Happened to the Six Million 2020 Voters?
Could Biden's votes in 2020 have included “ghost voters,” raising questions about the integrity of that election?
South Africans go to the polls
South Africans headed to the polls from early this morning.
Judicial Candidacy and the Judas Goat
If it smells like kak, look about for a leaking sewer. While DCJ Zondo is trying to crack the vault of state capture, there seems to be another capture right within the judiciary? Would we need another DCJ to investigate?
IT IS UMJOVO TIME!
A lying fake professor from a fake organization with fake members attempts to sway the undecided on vaccines through falsehood and share in the vaccine loot.
Broken New Year’s Resolution
Happy New 2021. Having resolved earlier this morning neither write nor speak anything about Donald Trump this year, I must apologize for failing so soon. I just couldn’t help it.
It's Joe
For five years, President Donald Nobody-ever-done-this-before Trump has succeeded in hypnotizing some American voters. Would the hypnosis effect end any time soon? With his present claims of electoral mass fraud without evidence.…
Is President Donald J Trump a Racist?
An easy task, crowning Coronavirus President of the year. it has been a slam dunk. Our countries that he once called shithole, have somehow managed to maintain relatively lower numbers of Cory deaths than Trump's America. And…
The unAwarded Nobel Prize
• The lead author of pseudo-scientific-claims-without-evidence has instituted a defamation lawsuit against our Editor, Mark Thomas • The suit doesn't worry us because all the statements on this website are based on facts. •…
FIDENTIA. WHO WATCHES THE WATCHERS
In South Africa, rarely do entities come out of curatorship smelling of roses, the appointed curators/liquidators (mostly men in suits and matching ties; like the disgraced Dines Gihwala) often come with trepidation among stakeholders.…
Tokyo Sexwale, Were the Guptas Worth It?
After lying down with dogs for over a decade, does the former Apprentice SA host want the public to believe he is not now getting up without fleas? Like his American Apprentice counterpart, Tokyo Sexwale seems to know so well…
Disruptive Developers
In the scramble for the most precious resource – water – ‘some animals are more equal than others’. Municipalities are left unable to plan adequately as some grab more than their authorised shares of services.
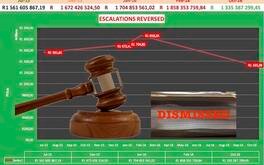
Rogue Tenderers: Treasury Drops the Gavel
Suppliers of pharmaceutical products are put on notice as Treasury moves to close loopholes in public procurement
State of Capture
The office of the Public Protector has released the much-anticipated report on allegations of state capture
The Big Plunder of Resources
Resource-dependent developing countries are left heavily impoverished as trillions of dollars illicitly flow out. But who is looking for a plan to stop the haemorrhage?
Grabbing the Roots
In the seemingly lawless frontier of bioresources, squabbles over who has the right to profit from knowledge and wealth have escalated. Image: GreenLITers
Behind Kapa’s Multi-billion Rand Covert Sale
Following the money: - the aftermath of experts’ failure to identify the company’s ‘real estate’ during valuation. How many more were victimized during the divorce and subsequent acquisition?
Tenders, Corruption and Murder
A devious scheme that caused the death of a Port Elizabeth businessman has taken corruption in the windy city to new heights.
Tales From the Dead
Was Soonthra ‘Samoo’ Chetty’s pen mightier than a sword? He protested the injustices meted out to his workers and lost everything, including his life. His story should rile those who failed him.
Multi-Billion Rand Investment Rip-off
The sweet-talking Americans came, saw and swindled. If one thought revelations within the Panama-Papers were devastating, try that of the American goons.
Union’s Dirty Hands on Pension
Is Cosatu’s call for the scrapping of new tax laws affecting pensions really in the interest of workers? Perhaps it is time workers looked elsewhere for leadership.